Intracellular Trafficking
- Following profitable penetration inside cells, the virus particles must get to an applicable website within the cell for genome
replication. This course of is termed intracellular trafficking. - The motion of nearly all of viruses inside the cell use microtubules however some viruses have been described to make use of actin cytoskeleton of filament.
- For viruses that replicate within the cytoplasm, the viral nucleocapsids have to be routed to the positioning for replication. These viruses use microtubule-mediated transport coupled with receptor-mediated endocytosis mechanism for the transport inside the cytoplasm. Clathrin-mediated endocytosis (essentially the most generally noticed uptake pathway for viruses), viruses are transported by way of the early endosome to the late endosome and finally to the lysosome. Within the Caveolin/lipid-raft mediated endocytosis, brings viruses to caveosomes. By the second vesicle transport step, viruses are transported to Golgi, after which to Endoplasmic Reticulum (ER).

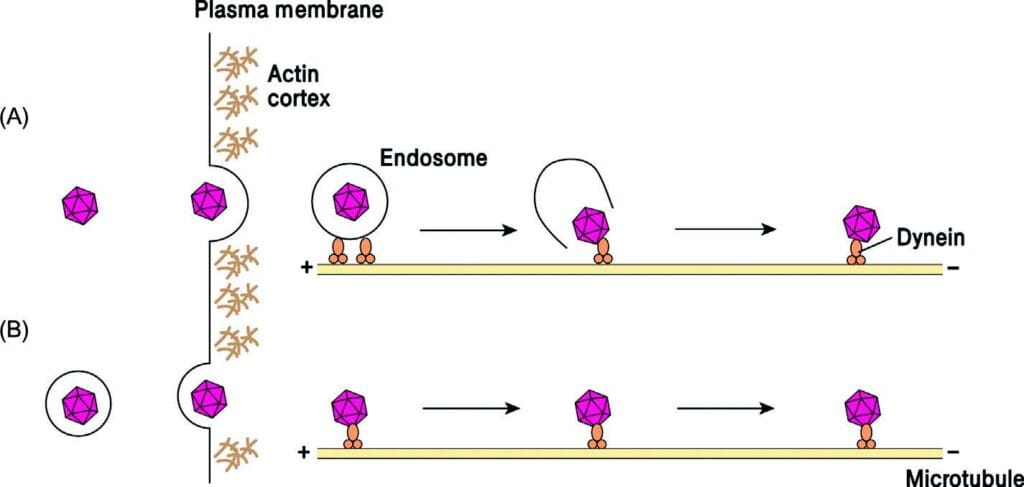
Cytoplasmic trafficking. Two distinct viruses are used to clarify how the entry is linked to cytoplasmic trafficking: (A) adenovirus
(bare) and (B) herpes virus (enveloped). Incoming viruses can enter cells by endocytosis (A) or direct fusion (B). Following penetration into cytoplasm, both endocytic vesicles or viral capsids exploit dynein motors to visitors towards the minus ends of microtubules. Both the endocytic vesicles (A) or the capsids (B) work together immediately with the microtubules. The virus can even lyse the endocytic membrane, releasing the capsid into the cytosol (A).
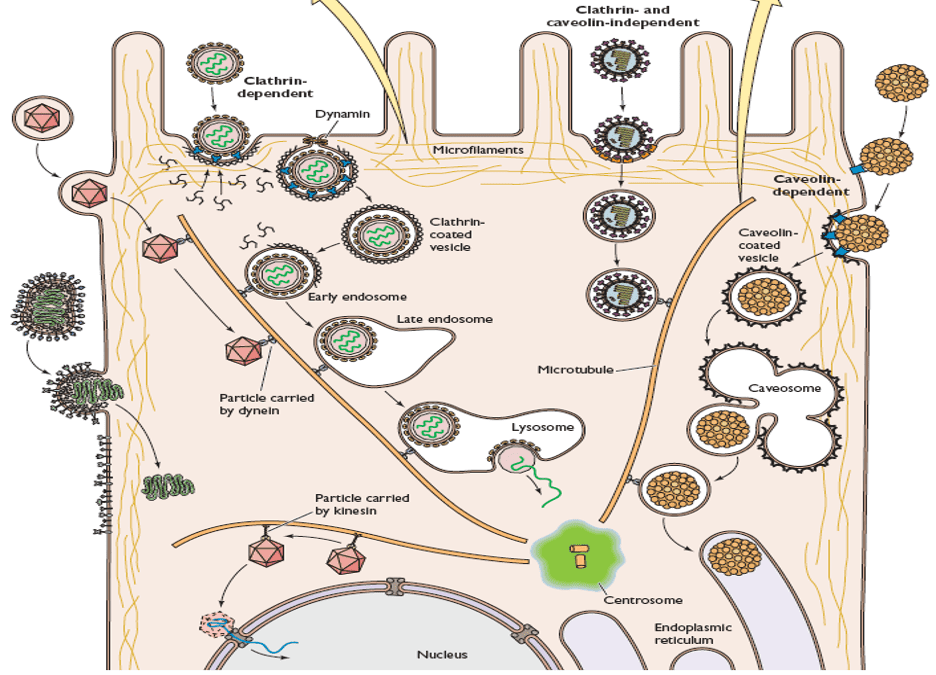
- For viruses that replicate within the nucleus, the viral nucleocapsids must enter the nucleus. For a lot of DNA viruses, the viral nucleocapsids are routed to the perinuclear space by way of microtubule-mediated transport. On this course of, a dynein motor powers the motion of virus particles. As an analogy, the viral nucleocapsids will be envisioned as a prepare in a railroad.
Cell-to-Cell Movement of Plant Viruses
- Cell-to-cell movement is a crucial step in plant virus infection.
- Virus cell-to-cell movement involves transport from replication factories to the cell periphery, passage through plasmodesmata (PD) channels interconnecting adjacent cells, and long-distance transport via the phloem vasculature.
- Plasmodesmata are lined with proteins and can be tightly controlled by the plant. Relative to the diameter of plasmodesmata, virus particles are huge. Imagine it’s like trying to pull a cat through a keyhole!
- Therefore, all plant viruses encode one or more special proteins, called movement proteins (MP), dedicated proteins to enlarging the pore size of plasmodesmata and actively transporting the viral nucleic acid into the adjacent cell, thereby allowing local and systemic spread of viruses in plants.
- The structurally and mechanistically diverse MP employ at least three different movement strategies.
- -(a) MP that directly binds and chaperones viral RNA genome via modified PD (represented by Tobacco mosaic virus).
- -(b) MP that heavily modify PD structure by forming tubules through which the assembled virions traverse PD.
- -(c) Usually require more than one MP and capsid protein for efficient intercellular transport (used primarily by the filamentous viruses such as closteroviruses).

References
- Wang-Shick Ryu. 2017. Molecular Virology of Human Pathogenic Viruses. Academic Press is an imprint of Elsevier
- Rojas, M.R. et al. (2016). Cell-to-Cell Movement of Plant Viruses: A Diversity of Mechanisms and Strategies. In: Wang, A., Zhou, X. (eds) Current Research Topics in Plant Virology. Springer, Cham. https://doi.org/10.1007/978-3-319-32919-2_5
- Amari K, Lerich A, Schmitt-Keichinger C, Dolja VV, Ritzenthaler C. Tubule-guided cell-to-cell movement of a plant virus requires class XI myosin motors. PLoS Pathog. 2011 Oct;7(10):e1002327. doi: 10.1371/journal.ppat.1002327. Epub 2011 Oct 27. PMID: 22046131; PMCID: PMC3203191.